
PATIENT CASES
VITRAKVI®: EXPLORE DISTINCT ADULT AND PEDIATRIC PATIENT CASES ACROSS SOLID TUMORS1-5
Each individual case reports a response from 1 patient. Different patients have different responses to therapy. To see efficacy by solid tumor type from our Initial Data Set, click here.
Explore the treatment journey of actual VITRAKVI patients. Each case provides an overview inclusive of the patient’s clinical presentation, previous treatments, molecular testing profile, VITRAKVI treatment regimen, and disease resolution.
Click below to choose a patient case by disease profile.
NSCLCa PATIENTS WITH METASTATIC DISEASE
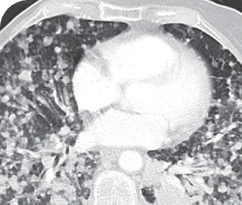
BASELINE
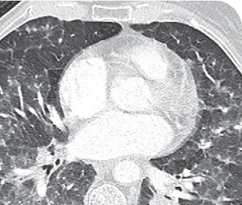
MONTH 4 OF TREATMENT
76-YEAR-OLD FEMALE WITH BRAIN METASTASES1
Patient Case - 76 year old - landing
PATIENT CASE:
NSCLCa PATIENT WITH BRAIN
METASTASES1-3
- 76-year-old female with NSCLC that metastasized to the brain and liver
- No prior systemic treatment before trial enrollment
- PRa of primary lung nodules to VITRAKVI® (larotrectinib) confirmed at 2 months1
This is a response from 1 patient. Different patients may have different responses to therapy.
This patient profile is adapted from a real patient case.
Clinical presentation1,2
- Presented with a persistent cough, fatigue, and anorexia
- Imaging identified bilateral lung nodules with metastases to the brain
- Endobronchial biopsy confirmed lung adenocarcinoma
Prior treatments and outcomes3
- No prior surgery or radiation
- Patient elected not to undergo platinum doublet chemotherapy treatment for her lung cancer in front line
aNSCLC, non-small cell lung cancer; PR, partial response.
Advise women not to breastfeed during treatment with VITRAKVI and for 1 week after the final dose.4
Testing1
- Initial DNAa-based NGSa testing was negative for NTRKa gene fusions
- Subsequent RNAa-based NGS testing identified EPS15a-NTRK1 gene fusion
NCCNa Clinical Practice Guidelines in Oncology (NCCN Guidelines®) recommend molecular testing that includes NTRK gene fusion for advanced or metastatic NSCLC.5
aDNA, deoxyribonucleic acid; EPS15, epidermal growth factor receptor pathway substrate; NCCN, National Comprehensive Cancer Network® (NCCN®); NGS, next-generation sequencing; NSCLC, non-small cell lung cancer; NTRK, neurotrophic receptor tyrosine kinase; PR, partial response; RNA, ribonucleic acid.
VITRAKVI treatment2
- VITRAKVI 100 mg twice daily orally
Response to VITRAKVI1,2
- Partial response to treatment that was confirmed at 2 months (34% tumor reduction)
- Grade 1 cough and Grade 2 fatigue
- Patient remains on VITRAKVI after more than 4 months of treatment
aNSCLC, non-small cell lung cancer; PR, partial response.
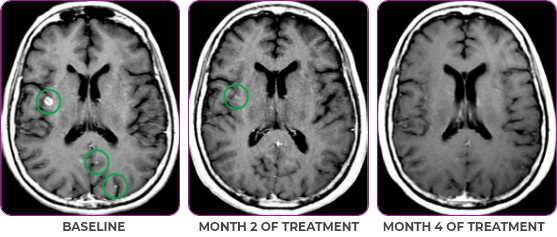
Response in primary and metastatic lesions1
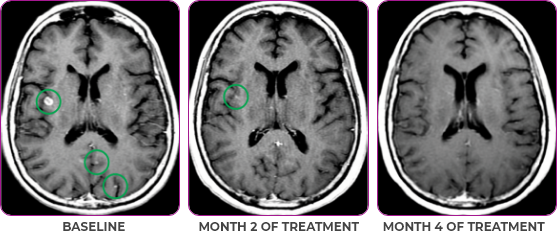
MRIa imagery of the brain. Green circles indicate baseline brain metastases.1

Response in primary and metastatic lesions1
SCAN 2: BRAIN
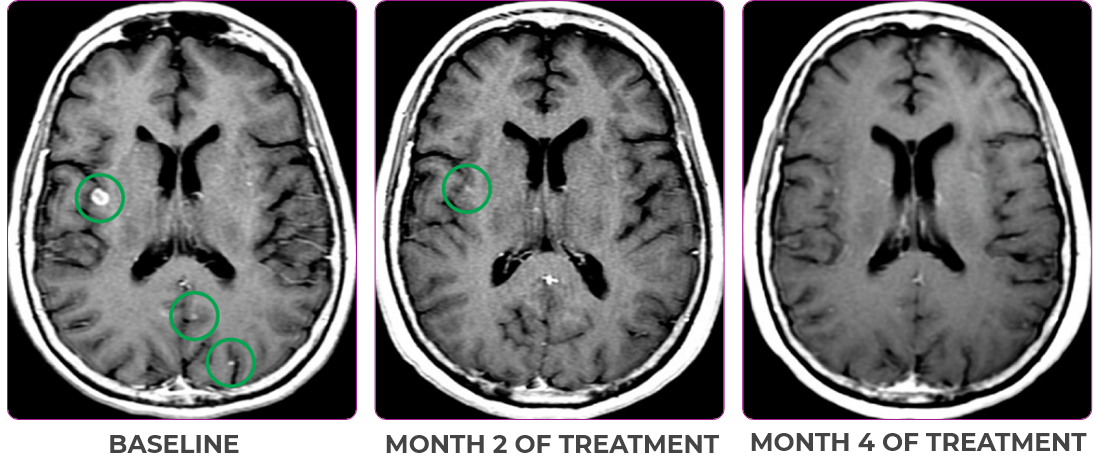
MRIa imagery of the brain. Green circles indicate baseline brain metastases.1
aMRI, magnetic resonance imaging.
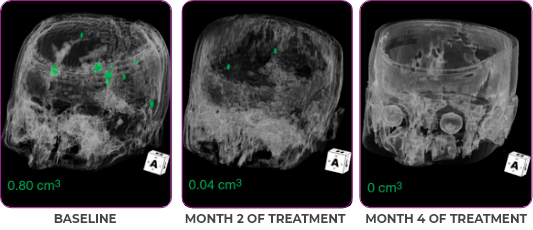
MRIa imagery of the brain. Green voxels indicate decreased burden of metastatic disease; further quantified by accompanying measurement in green.1

Response in primary and metastatic lesions1
SCAN 3: BRAIN
MRIa imagery of the brain. Green voxels indicate decreased burden of metastatic disease; further quantified by accompanying measurement in green.1
aMRI, magnetic resonance imaging.
Pre- and post-treatment imaging, by Rosen EY et al, is licensed under Creative Commons License CC BY 4.0.
aNSCLC, non-small cell lung cancer; PR, partial response.
References
- Rosen EY, Schram AM, Young RJ, et al. Larotrectinib demonstrates CNS efficacy in TRK fusion-positive solid tumors. JCO Precis Oncol. 2019;3:1-5. Return to content
- Lassen U, Albert CM, Kummar S, et al. Larotrectinib efficacy and safety in TRK fusion cancer: an expanded clinical dataset showing consistency in an age and tumor agnostic approach. Presented at: European Society for Medical Oncology Congress; October 19-23, 2018; Munich, Germany. Return to content
- Drilon A, Moreno V, Patel J, et al. Efficacy and safety of larotrectinib in patients with tropomyosin receptor kinase (TRK) fusion lung cancer. Poster presented at: ESMO Virtual Congress 2020; September 19-21, 2020. Return to content
- VITRAKVI [package insert]. Whippany, NJ: Bayer HealthCare Pharmaceuticals, Inc.; April 2025. Return to content
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Non-Small Cell Lung Cancer V.7.2024. © National Comprehensive Cancer Network, Inc. 2024. All rights reserved. Accessed July 26, 2024. To view the most recent and complete version of the guidelines, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. Return to content
aNSCLC, non-small cell lung cancer; PR, partial response.
PATIENT CASE:
NSCLCa PATIENT WITH BRAIN
METASTASES1-3
- 76-year-old female with NSCLC that metastasized to the the brain and liver
- No prior systemic treatment before trial enrollment
- PRa of primary lung nodules to VITRAKVI® (larotrectinib) confirmed at 2 months1
This is a response from 1 patient. Different patients may have different responses to therapy.
This patient profile is adapted from a real patient case.
Clinical presentation1,2
- Presented with a persistent cough, fatigue, and anorexia
- Imaging identified bilateral lung nodules with metastases to the brain
- Endobronchial biopsy confirmed lung adenocarcinoma
Prior treatments and outcomes3
- No prior surgery or radiation
- Patient elected not to undergo platinum doublet chemotherapy treatment for her lung cancer in front line
aNSCLC, non-small cell lung cancer; PR, partial response.
Advise women not to breastfeed during treatment with VITRAKVI and for 1 week after the final dose.4
Testing1
- Initial DNAa-based NGSa testing was negative for NTRKa gene fusions
- Subsequent RNAa-based NGS testing identified EPS15a-NTRK1 gene fusion
NCCNa Clinical Practice Guidelines in Oncology (NCCN Guidelines®) recommend molecular testing that includes NTRK gene fusion for advanced or metastatic NSCLC.5
aDNA, deoxyribonucleic acid; EPS15, epidermal growth factor receptor pathway substrate; NCCN, National Comprehensive Cancer Network® (NCCN®); NGS, next-generation sequencing; NSCLC, non-small cell lung cancer; NTRK, neurotrophic receptor tyrosine kinase; PR, partial response; RNA, ribonucleic acid.
VITRAKVI treatment2
- VITRAKVI 100 mg BIDa orally
Response to VITRAKVI1,2
- Partial response to treatment that was confirmed at 2 months (34% tumor reduction)
- Grade 1 cough and Grade 2 fatigue
- Patient remains on VITRAKVI after more than 4 months of treatment
aBID, twice daily; NSCLC, non-small cell lung cancer; PR, partial response.
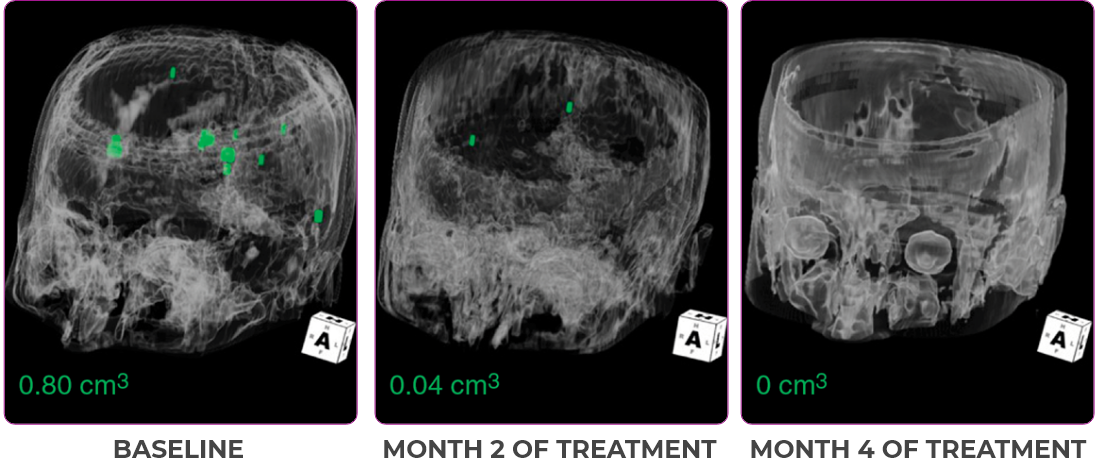
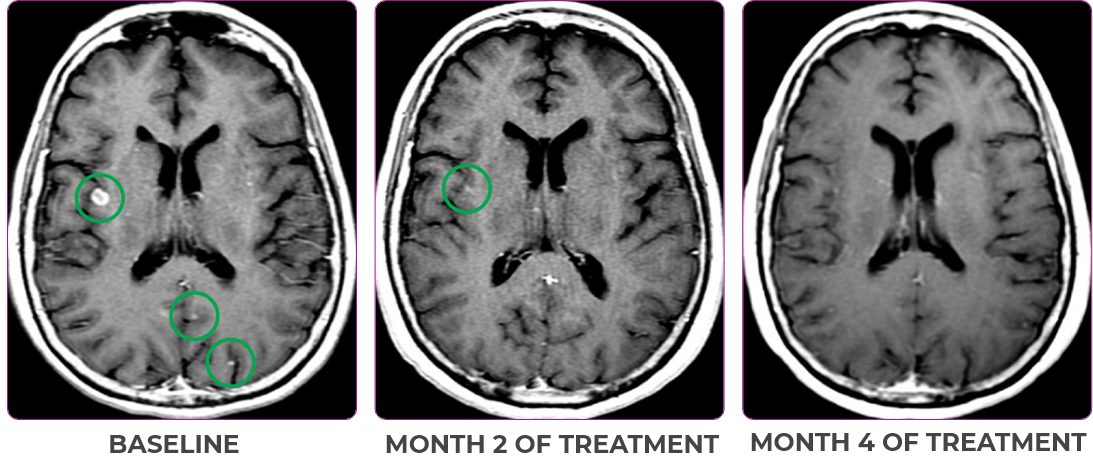
Response in primary and metastatic lesions1
MRIa imagery of the brain. Green circles indicate baseline brain metastases.1




Response in primary and metastatic lesions1
SCAN 2: BRAIN
MRIa imagery of the brain. Green circles indicate baseline brain metastases.1
aMRI, magnetic resonance imaging.
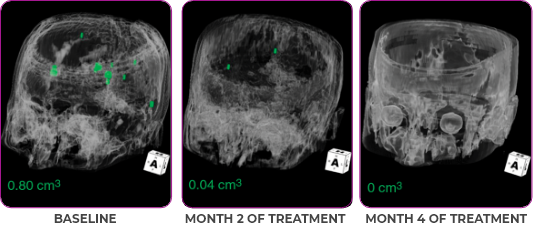
MRIa imagery of the brain. Green voxels indicate decreased burden of metastatic disease; further quantified by accompanying measurement in green.1

Response in primary and metastatic lesions1
SCAN 3: BRAIN
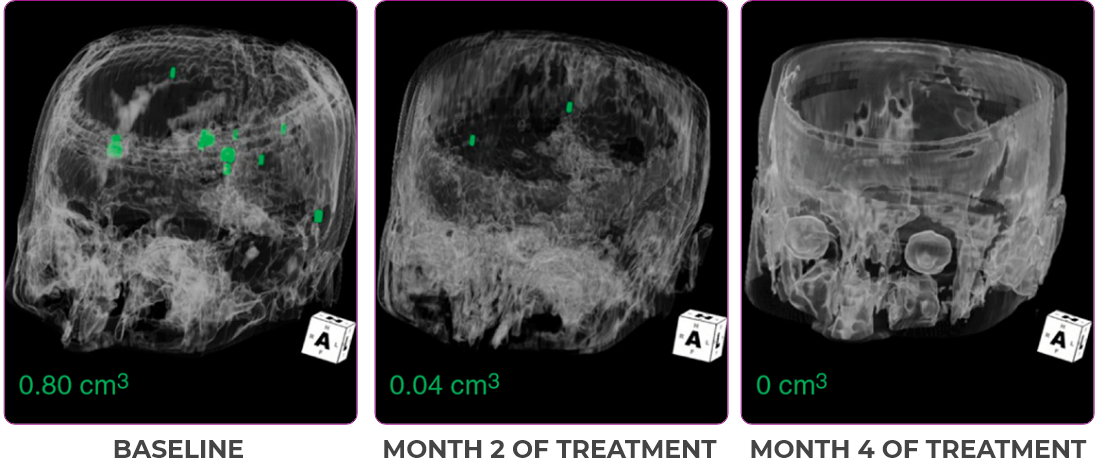
MRIa imagery of the brain. Green voxels indicate decreased burden of metastatic disease; further quantified by accompanying measurement in green.1
aMRI, magnetic resonance imaging.
Pre- and post-treatment imaging, by Rosen EY et al, is licensed under Creative Commons License CC BY 4.0.
aMRI, magnetic resonance imaging; NSCLC, non-small cell lung cancer; PR, partial response.
References
- Rosen EY, Schram AM, Young RJ, et al. Larotrectinib demonstrates CNS efficacy in TRK fusion-positive solid tumors. JCO Precis Oncol. 2019;3:1-5. Return to content
- Lassen U, Albert CM, Kummar S, et al. Larotrectinib efficacy and safety in TRK fusion cancer: an expanded clinical dataset showing consistency in an age and tumor agnostic approach. Presented at: European Society for Medical Oncology Congress; October 19-23, 2018; Munich, Germany. Return to content
- Drilon A, Moreno V, Patel J, et al. Efficacy and safety of larotrectinib in patients with tropomyosin receptor kinase (TRK) fusion lung cancer. Poster presented at: ESMO Virtual Congress 2020; September 19-21, 2020. Return to content
- VITRAKVI [package insert]. Whippany, NJ: Bayer HealthCare Pharmaceuticals, Inc.; December 2022. Return to content
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Non-Small Cell Lung Cancer V1.2023. © National Comprehensive Cancer Network, Inc. 2023. All rights reserved. Accessed January 4, 2023. To view the most recent and complete version of the guidelines, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. Return to content
aNSCLC, non-small cell lung cancer; PR, partial response.
Pre- and post-treatment imaging, by Rosen EY et al, is licensed under Creative Commons License CC BY 4.0.
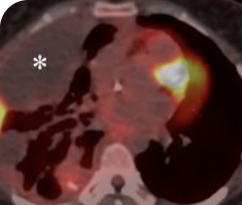
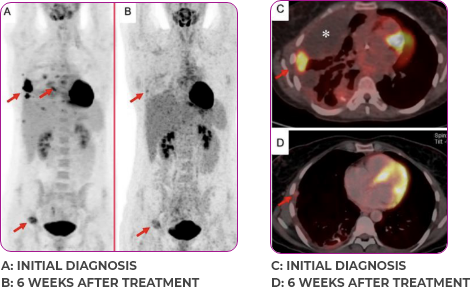
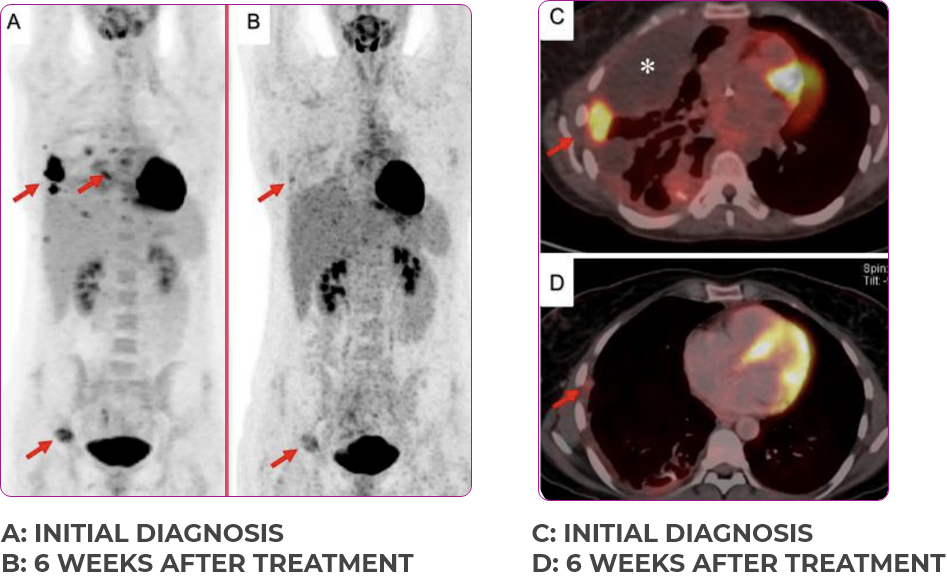
INITIAL DIAGNOSIS
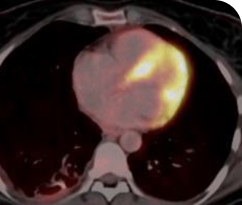
6 WEEKS AFTER TREATMENT
30-YEAR-OLD FEMALE WITH BONE METASTASES2
PATIENT CASE:
METASTATIC NSCLC1,a
- 30-year-old female
- Progression on first-line doublet chemotherapy
- PRa after 6 weeks of treatment with VITRAKVI® (larotrectinib)
- Complete clinical response achieved by 12 months with residual scarring
This is a response from 1 patient. Different patients may have different responses to therapy.
This patient profile is adapted from a real patient case.
Clinical presentation1
- Nonsmoker and a history of atopic eczema
- Presented with dyspnea and pleural effusion
- Bone-scan imaging revealed 3 metastatic lesions of the pelvis, in the sacrum, acetabulum, and ischial bones, which were asymptomatic
- Diagnosed with metastatic adenocarcinoma of the right lung in March 2017
Prior treatments and outcomes1
- 4 cycles of cisplatin plus pemetrexed, followed by pemetrexed maintenance for 1 year. Partial remission was achieved and treatment was discontinued at patient request
- Progression recurred 5 months later
- lmmunotherapy was considered, but not selected due to patient’s concern about autoimmune AEsa from preexisting atopic eczema
aAE, adverse event; NSCLC, non-small cell lung cancer; PR, partial response.
Advise women not to breastfeed during treatment with VITRAKVI and for 1 week after the final dose.2
Testing1
- NGSa testing was not routine at the time of diagnosis. Instead, molecular testing for EGFRa mutations (real-time PCRa), ALK,a and ROS1a (IHCa) was performed and showed no targetable abnormalities. PD-L1a expression was detected in approximately 1% of tumor cellsb
- NTRKa testing was not performed until 3 months after disease progression. Patient was found to be NTRK gene fusion–positive based on both IHC and NGS. TPM3a-NTRK1 gene fusion was identified as the oncogenic driver
NCCNa Clinical Practice Guidelines in Oncology (NCCN Guidelines®) recommend molecular testing that includes NTRK gene fusion for advanced or metastatic NSCLC.3
aALK, anaplastic lymphoma kinase; EGFR, epidermal growth factor receptor; FDA, US Food and Drug Administration; IHC, immunohistochemistry; NCCN, National Comprehensive Cancer Network® (NCCN®); NGS, next-generation sequencing; NSCLC, non-small cell lung cancer; NTRK, neurotrophic receptor tyrosine kinase; PCR, polymerase chain reaction; PD-L1, programmed death-ligand 1; PR, partial response; ROS1, c-ros oncogene 1; TPM3, tropomyosin 3.
bSelect patients for therapy based on FDAa-approved test.1
VITRAKVI treatment1
- VITRAKVI 100 mg twice daily orally
Response to VITRAKVI1
- Partial response and symptom improvement confirmed by chest X-rays after <1 month of treatment
- Imaging performed 6 weeks into treatment revealed considerable decrease in the size of both target lesions
- No treatment-related AEsa were reported
- Complete clinical response achieved by 12 months with residual scarring
- Patient was symptom free and remained on VITRAKVI
aAE, adverse event; NSCLC, non-small cell lung cancer; PR, partial response.
Response in primary and metastatic lesions1
Lung imaging of primary tumors.
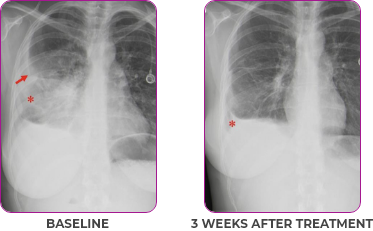

Response in primary and metastatic lesions1
Lung imaging of primary tumors.
SCAN 1: LUNG
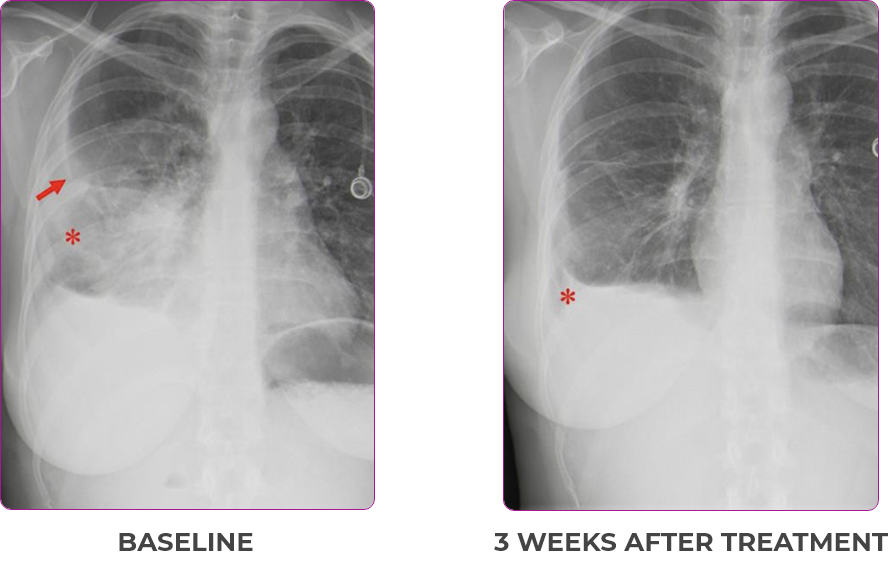

Response in primary and metastatic lesions1
Lung imaging of primary tumors.
SCAN 2: LUNG AND BONE
Images courtesy of Dr Maximilian Hochmair.
aNSCLC, non-small cell lung cancer; PR, partial response.
References
- Blay J-Y, Brose MS, García-Foncillas J, Hochmair MJ, Vainer G. Precision medicine in real-world clinical practice: perspectives from a multidisciplinary team. Virtual presentation at: European Society for Medical Oncology Virtual Congress 2020 Industry Satellite Symposium. On demand from September 14, 2020. Return to content
- VITRAKVI [package insert]. Whippany, NJ: Bayer HealthCare Pharmaceuticals, Inc.; April 2025. Return to content
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Non-Small Cell Lung Cancer V.7.2024. © National Comprehensive Cancer Network, Inc. 2024. All rights reserved. Accessed July 26, 2024. To view the most recent and complete version of the guidelines, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. Return to content
aNSCLC, non-small cell lung cancer; PR partial response.
Images courtesy of Dr Maximilian Hochmair.
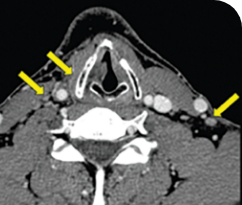
THYROID CANCER PATIENTS WITH METASTATIC DISEASE
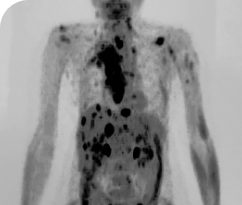
BASELINE

AFTER 2 MONTHS OF TREATMENT
56-YEAR-OLD FEMALE WITH MULTIPLE METASTASES3
PATIENT CASE:
METASTATIC, RAIa-REFRACTORY,
PAPILLARY THYROID CANCER1
- 56-year-old female with PTCa that had metastasized to multiple sites, including the brain
- Progression after first- and second-line systemic treatment
- PRa after 4 weeks on VITRAKVI® (larotrectinib); CRa after 8 weeks
- At last assessment, complete response was sustained over an 11-month period
This is a response from 1 patient. Different patients may have different responses to therapy.
This patient profile is adapted from a real patient case.
Clinical presentation1
- Patient was assessed to have metastatic, rapidly progressive, RAl-refractory PTC based on imaging that revealed multiple metastatic sites, including the neck, mediastinum, lung, scalp, and bones
- Biopsy of a scalp lesion helped to confirm the PTC metastasis
- Metastases to the brain and liver were observed after progression on systemic treatments
Prior treatments and outcomes1
- Prior treatments included surgery, RAI, a combination of zoledronic acid and sorafenib, external beam radiotherapy, and then lenvatinib
aCR, complete response; PR, partial response; PTC, papillary thyroid cancer; RAI, radioactive iodine.
Advise women not to breastfeed during treatment with VITRAKVI and for 1 week after the final dose.2
Testing1
- With no further treatment options, the decision was made to perform genomic testing. NGSa testing was performed on a scalp biopsy and revealed an ETV6a-NTRK3a gene fusion. The scalp biopsy had been performed 1 year earlier.
NCCNa Clinical Practice Guidelines in Oncology (NCCN Guidelines®) recommend genomic testing to identify actionable mutations, including NTRK, for advanced, progressive, or threatening thyroid carcinoma.3
aCR, complete response; ETV6, ETS variant 6; NCCN, National Comprehensive Cancer Network® (NCCN®); NGS, next-generation sequencing; NTRK, neurotrophic receptor tyrosine kinase; PR, partial response; PTC, papillary thyroid cancer; RAI, radioactive iodine.
VITRAKVI treatment1
- VITRAKVI 100 mg twice daily orally
Response to VITRAKVI1
- After 8 weeks on VITRAKVI, all target lesions had disappeared, demonstrating a complete response
- The complete response was sustained over 11 months of treatment with VITRAKVI
- Patient experienced Grade 1 fatigue and mild hepatic enzyme elevation
aCR, complete response; PR, partial response; PTC, papillary thyroid cancer; RAI, radioactive iodine.
Response in primary and metastatic lesions1
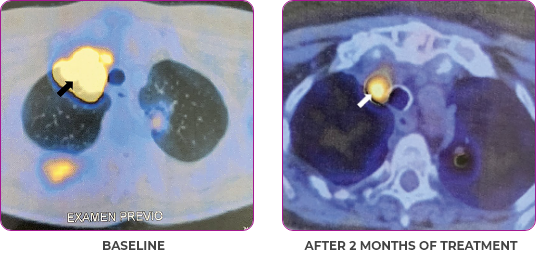
MRIa imagery of the lungs. Arrow indicates large target lesion in left lung.1

Response in primary and metastatic lesions1
SCAN 1: LUNG
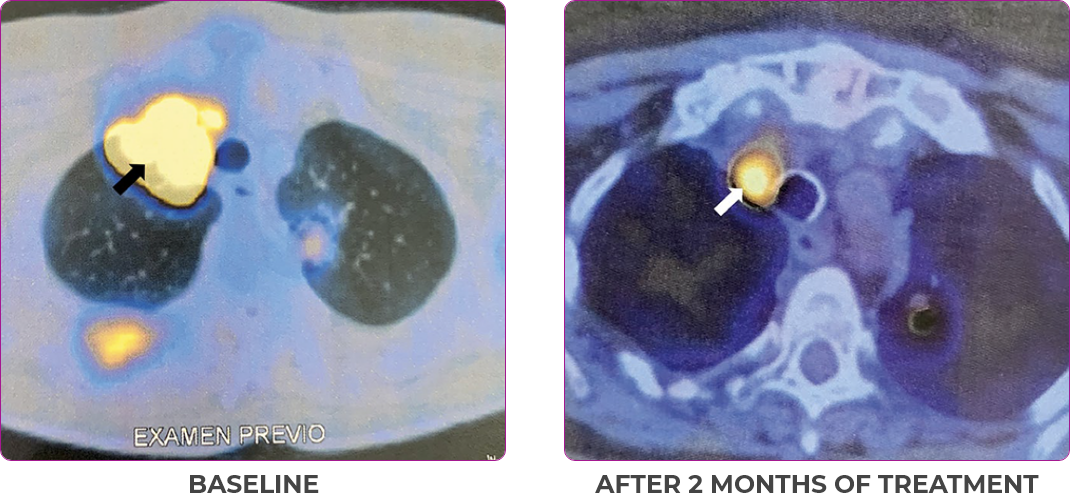
MRIa imagery of the brain. Arrow indicates large target lesion in left lung.1
aMRI, magnetic resonance imaging.
Images courtesy of Dr Fabian Pitoia.
aCR, complete response; MRI, magnetic resonance imaging; PR, partial response; PTC, papillary thyroid cancer; RAI, radioactive iodine.
References
- Pitoia F. Complete response to larotrectinib treatment in a patient with papillary thyroid cancer harboring an ETV6-NTRK3 gene fusion. Clin Case Rep. 2021;9(4):1905-1912. Return to content
- VITRAKVI [package insert]. Whippany, NJ: Bayer HealthCare Pharmaceuticals, Inc.; April 2025. Return to content
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Thyroid Carcinoma V.3.2024. © National Comprehensive Cancer Network, Inc. 2024. All rights reserved. Accessed July 26, 2024. To view the most recent and complete version of the guidelines, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. Return to content
aCR, complete response; PR, partial response; PTC, papillary thyroid cancer; RAI, radioactive iodine.
Images courtesy of Dr Fabian Pitoia.
BASELINE
AFTER 31 MONTHS
33-YEAR-OLD MALE4
PATIENT CASE:
METASTATIC, RAIa-REFRACTORY,
PAPILLARY THYROID CANCER1,2
- 33-year-old male
- Progression on RAI and first line systemic therapy
- PRa after 2 cycles of VITRAKVI® (larotrectinib)
- Confirmed partial response on VITRAKVI lasting 55 cycles
This is a response from 1 patient. Different patients may have different responses to therapy.
This patient profile is adapted from a real patient case.
Clinical presentation1
- Initially diagnosed at age 27
- Bulky disease in neck and thoracic involvement; metastatic, RAI-refractory papillary thyroid cancer
Prior treatments and outcomes1
- 5 prior surgeries
- 2 prior RAI treatments
- Systemic therapy with pazopanib and trametinib
aPR, partial response; RAI, radioactive iodine.
Testing1,2
- ETV6a-NTRK3a gene fusion was detected during disease progression on systemic therapy via NGS
NCCNa Clinical Practice Guidelines in Oncology (NCCN Guidelines®) recommend genomic testing to identify actionable mutations, including NTRK, for advanced, progressive, or threatening thyroid carcinoma.3
aETV6, ETS variant 6; NCCN, National Comprehensive Cancer Network® (NCCN®); NTRK, neurotrophic receptor tyrosine kinase; PR, partial response; RAI, radioactive iodine.
VITRAKVI treatment1
- VITRAKVI 100 mg twice daily orally
Response to VITRAKVI1,2
- Confirmed partial response (92.6% reduction in target lesions)
- Treatment continued for 55 cycles
- VITRAKVI was well tolerated; patient did not have to discontinue due to AEa
- No treatment interruptions or dose reductions
aAE, adverse event; PR, partial response; RAI, radioactive iodine.
Response in primary and metastatic lesions1
Images courtesy of Dr Steven Waguespack.
aPR, partial response; RAI, radioactive iodine.
References
- Waguespack SG, Drilon A, Farago AF, et al. Treatment of advanced TRK fusion thyroid cancer with larotrectinib. Presented at: 42nd Annual Meeting of the European Thyroid Association; September 7-10, 2019; Budapest, Hungary. Return to content
- Data on file. Bayer HealthCare Pharmaceuticals, Inc., Whippany, NJ. Return to content
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Thyroid Carcinoma V.3.2024. © National Comprehensive Cancer Network, Inc. 2024. All rights reserved. Accessed July 26, 2024. To view the most recent and complete version of the guidelines, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. Return to content
aPR, partial response; RAI, radioactive iodine.
Images courtesy of Dr Steven Waguespack.
PEDIATRIC CANCER PATIENT WITH METASTATIC DISEASE
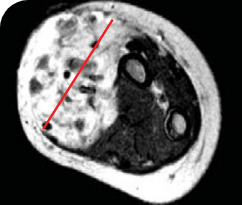
BASELINE BEFORE VITRAKVI
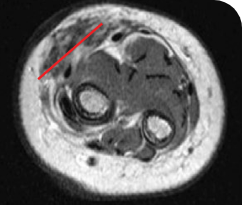
AFTER 6 MONTHS ON VITRAKVI
5-MONTH-OLD WITH INFANTILE FIBROSARCOMA5
PATIENT CASE:
INFANTILE FIBROSARCOMA
OF SOFT TISSUE1
- 5-month-old with IFSa
- Progression after chemotherapy
- Confirmed PRa after 4 cycles of VITRAKVI® (larotrectinib)
- Referred for definitive limb-sparing surgery after 6 cycles
of VITRAKVI; achieved pCRa,b
This is a response from 1 patient. Different patients may have different responses to therapy.
This patient profile is adapted from a real patient case.
Clinical presentation1
- Diagnosed with localized IFS of the soft tissue of the forearm
Prior treatments and outcomes1
- Patient progressed after 2 cycles of vincristine and actinomycin D
- He was subsequently placed on vincristine, actinomycin D, and cyclophosphamide; however, his response remained inadequate to allow for limb-sparing surgery
aIFS, infantile fibrosarcoma; pCR, pathological complete response; PR, partial response.
bPatients undergoing a surgical resection whose postoperative pathologic assessment showed no viable tumor cells and negative margins were pathological complete responders provided that no other sites of disease were present.2
Advise women not to breastfeed during treatment with VITRAKVI and for 1 week after the final dose.2
Testing1
- ETV6a-NTRK3a fusion identified with DNAa NGSa panel testing
aDNA, deoxyribonucleic acid; ETV6, ETS variant 6; IFS, infantile fibrosarcoma; NTRK, neurotrophic receptor tyrosine kinase; NGS, next-generation sequencing; pCR, pathological complete response; PR, partial response.
bPatients undergoing a surgical resection whose postoperative pathologic assessment showed no viable tumor cells and negative margins were pathological complete responders provided that no other sites of disease were present.2
VITRAKVI treatment1
- VITRAKVI 100 mg twice daily orally
Response to VITRAKVI1
- Patient achieved a confirmed partial response after 4 cycles of VITRAKVI with a 45% reduction in tumor burden
- Following 6 cycles of VITRAKVI, the patient was referred for definitive limb-sparing surgery
- Pathology revealed a complete pathologic response and clear resection margins with scar tissue noted
aIFS, infantile fibrosarcoma; pCR, pathological complete response; PR, partial response.
bPatients undergoing a surgical resection whose postoperative pathologic assessment showed no viable tumor cells and negative margins were pathological complete responders provided that no other sites of disease were present.2
Reduction in tumor burden1
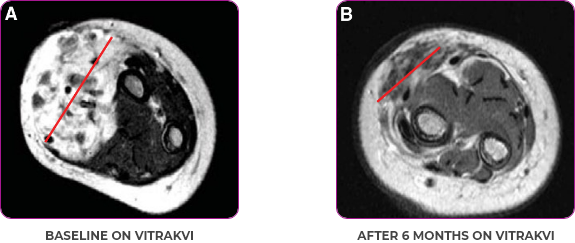
MRIa imagery of the brain. Red line indicates the maximum dimension.1

Reduction in tumor burden1
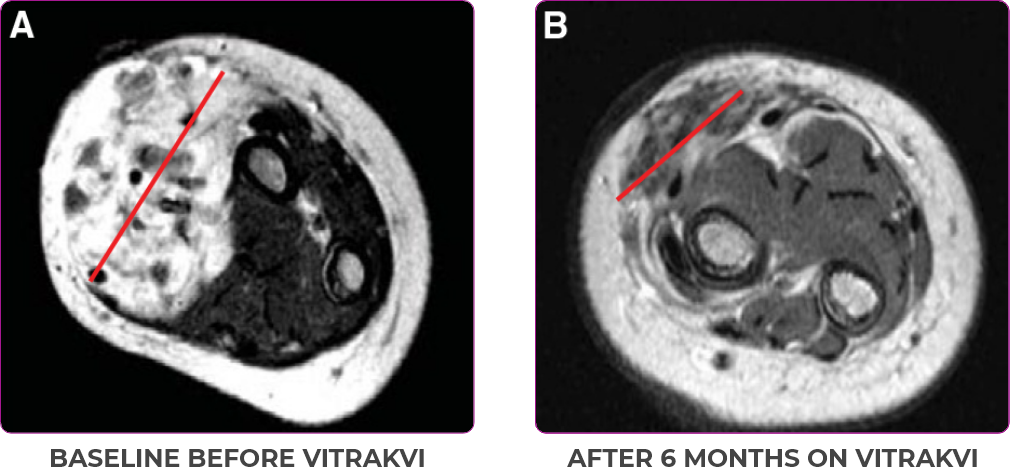
MRIa imagery of the brain. Red line indicates the maximum dimension.1
aMRI, magnetic resonance imaging.
Pre- and post-treatment imaging, by DuBois SG et al, is licensed under CC BY 4.0.
aIFS, infantile fibrosarcoma; MRI, magnetic resonance imaging; pCR, pathological complete response; PR, partial response.
bPatients undergoing a surgical resection whose postoperative pathologic assessment showed no viable tumor cells and negative margins were pathological complete responders provided that no other sites of disease were present.2
References
- DuBois SG, Laetsch TW, Federman N, et al. The use of neoadjuvant larotrectinib in the management of children with locally advanced TRK fusion sarcomas. Cancer. 2018;124(21):4241-4247. Return to content
- VITRAKVI [package insert]. Whippany, NJ: Bayer HealthCare Pharmaceuticals, Inc.; April 2025. Return to content
aIFS, infantile fibrosarcoma; pCR, pathological complete response; PR, partial response
bPatients undergoing a surgical resection whose postoperative pathologic assessment showed no viable tumor cells and negative margins were pathological complete responders provided that no other sites of disease were present.2
Pre- and post-treatment imaging, by Rosen EY et al, is licensed under Creative Commons License CC BY 4.0.
aNSCLC, non-small cell lung cancer; NTRK, neurotrophic receptor tyrosine kinase.
References
- Rosen EY, Schram AM, Young RJ, et al. Larotrectinib demonstrates CNS efficacy in TRK fusion-positive solid tumors. JCO Precis Oncol. 2019;3:1-5. Return to content
- Blay J-Y, Brose MS, García-Foncillas J, Hochmair MJ, Vainer G. Precision medicine in real-world clinical practice: perspectives from a multidisciplinary team. Virtual presentation at: European Society for Medical Oncology Virtual Congress 2020 Industry Satellite Symposium. On demand from September 14, 2020. Return to content
- Pitoia F. Complete response to larotrectinib treatment in a patient with papillary thyroid cancer harboring an ETV6-NTRK3 gene fusion. Clin Case Rep. 2021;9(4):1905-1912. Return to content
- Waguespack SG, Drilon A, Farago AF, et al. Treatment of advanced TRK fusion thyroid cancer with larotrectinib. Presented at: 42nd Annual Meeting of the European Thyroid Association; September 7-10, 2019; Budapest, Hungary. Return to content
- DuBois SG, Laetsch TW, Federman N, et al. The use of neoadjuvant larotrectinib in the management of children with locally advanced TRK fusion sarcomas. Cancer. 2018;124(21):4241-4247. Return to content































